Christo Sevov
Associate Professor
186 CBEC Building
151 W Woodruff Ave
Columbus, OH 43210
Areas of Expertise
- Organic
Bio
Christo Sevov was born in Sofia, Bulgaria and moved to the US at a young age. He spent his formative years in South Bend, IN and received his B.S. in 2009 from the University of Notre Dame. There, Christo worked with Prof. Olaf Wiest on photocatalyzed cycloaddition reactions with selectivities that complement those of traditional Diels-Alder processes. Christo earned his Ph.D. in 2014 after beginning his studies at the University of Illinois Urbana-Champaign with Prof. John Hartwig and later moving with the group to the University of California Berkeley. His research involved the development of methods and mechanistic study of metal-catalyzed additions of C–H, N–H, and O–H bonds across alkenes. Following his doctoral studies, Christo conducted his postdoctoral work with Prof. Melanie Sanford at the University of Michigan. Applying an organic chemist’s approach to energy storage, Christo helped establish a new research program aimed towards the design of solvated battery materials for redox flow batteries. Christo is joining the faculty of The Ohio State University in July 2017 as an assistant professor in the Department of Chemistry and Biochemistry.
Research Overview
Research in the Sevov lab aims to develop strategies at the interface of homogeneous catalysis and electrochemistry for the sustainable utilization of electrical energy that is generated from renewable sources. By treating electrical energy as a reagent, our group aims to demonstrate that toxic, explosive, or expensive reagents, which drive traditional synthetic organic processes, can be replaced with inexpensive and benign additives, thereby enabling organic synthesis to be performed in a safe, sustainable, and scalable manner. In addition to the application of electrical energy to synthesis, we seek to design large scale energy storage systems that can assist with the integration of intermittent electrical loads from renewable sources into the electrical grid. Sevov lab members addressing these challenges will be trained and exposed to the multidisciplinary fields of organic synthesis, catalyst design, mechanistic study, materials development, and electroanalytical evaluation.
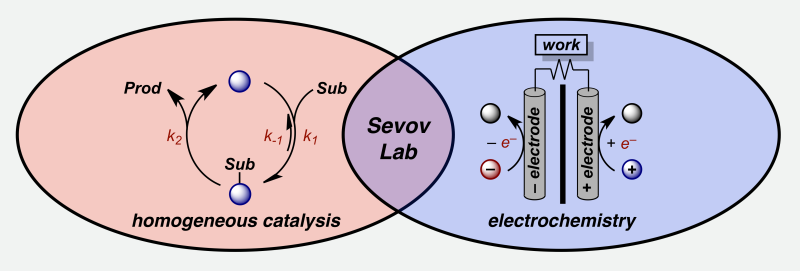
Electrically-Driven Homogeneous Catalysis
The development of metal-catalyzed reactions has revolutionized synthetic organic chemistry. Because many of these catalytic processes necessitate changes to the oxidation state of the metal, catalysts are generally of noble metals with vetted ligands that facilitate the redox events. To render these transformations favorable, high energy additives are added or substrates are prefunctionalized. Our lab is interested in utilizing electrochemistry in combination with homogeneous catalysis to drive chemical reactions by modulating the input of electrical energy, which allows electrons to be treated like inexpensive reagents. In doing so, our lab will develop methods for complex organic synthesis that (i) utilize earth-abundant metal catalysts, (ii) convert stoichiometric reagents into catalysts, and (iii) access high-energy intermediates under mild conditions to accelerate turnover-limiting processes.
Valorization of Abundant Feedstocks and Wastes
The accumulation of chemical waste products and the increasing need for new sources of commodity chemicals invites exciting opportunities for creative solutions to these global challenges. While conventional methods for upgrading or recycling wastes to desirable commodity chemicals require the use of harsh or expensive reagents, our lab will utilize electrical energy as a thermodynamic driving force for these processes. With this strategy, we will develop sustainable methods for (i) valorizing biofeedstocks, (ii) capturing greenhouse gases, and (iii) recycling wastes such as chlorofluorocarbons (CFCs).
Large-Scale Energy Storage
Integration of electrical energy that is harnessed by wind turbines or photovoltaics into the electrical grid requires large-scale energy storage systems that can modulate the variable and intermittent nature of these energy sources. However, the immense scale of this challenge precludes the use of most established battery technologies due to their cost. Our group will pursue an interdisciplinary approach of organic synthesis, polymers and materials development, and electroanalytical chemistry to invent scalable energy storage devices that utilize inexpensive liquid anode and cathode materials for shuttling and storing energy.
The Sevov lab is currently looking for highly motivated graduate and undergraduate students with interests in organic and organometallic synthesis, catalysis, or energy storage. Inquiries about positions in the group are welcomed.
Selected Publications
General C(sp2)-C(sp3) Cross-Electrophile Coupling Reactions Enabled by Overcharge Protection of Homogeneous Electrocatalysts. Truesdell, B. L; Hamby, T. B.; Sevov, C. S., J. Am. Chem. Soc. 2020, 142, 5884.
Direct and Scalable Electroreduction of Triphenylphosphine Oxide to Triphenylphosphine. Manabe, S.; Wong, C. M.; Sevov, C. S., J. Am. Chem. Soc. 2020, 142, 3024.
(Selected as JACS Cover, Write-up in Chemical Engineering magazine, Press release on Electrifying Aluminum, Highlighted by the Sustainability Institute)
An Electrochemically-Promoted, Nickel-Catalyzed, Mizoroki-Heck Reaction. Walker, B. R.; Sevov, C. S., ACS Catal. 2019, 9, 7197.
Effect of the Backbone Tether on the Electrochemical Properties of Soluble Cyclopropenium Redox-Active Polymers. Montoto, E. C.; Cao, Y.; Hernández-Burgos, K.; Sevov, C. S.; Braten, M. N.; Helms, B. A.; Moore, J. S.; Rodríguez-López, J., Macromolecules 2018, 10, 3539.
(Selected as Journal Cover)
High-Performance Oligomeric Catholytes for Effective Macromolecular Separation in Nonaqueous Redox Flow Batteries. Hendriks, K. H.; Robinson, S. G.; Braten, M. N.; Sevov, C. S.; Helms, B. A.; Sigman, M. S.; Minteer, S. D.; Sanford, M. S. ACS Cent. Sci. 2018, 4, 189.
Sevov, C. S.; Hickey, D. P.; Cook, M. E.; Robinson, S. G.; Barnett, S.; Minteer, S. D.; Sigman, M. S.; Sanford, M. S., “Physical Organic Approach to Persistent, Cyclable, Low-Potential Anolytes for Flow Battery Applications” J. Am. Chem. Soc., 2017, ASAP, DOI: 10.1021/jacs.7b00147.
Sevov, C. S.; Samaroo, S. K.; Sanford, M. S.; “Cyclopropenium Salts as Cyclable, High-Potential Catholytes in Non-Aqueous Media” Adv. Energy Mater. 2016, 1602027.
Sevov, C. S.; Laramie, S. M.; Thompson, L. T.; Sanford, M. S., “Mechanism-Based Development of a Low-Potential, Soluble, and Cyclable Anolyte for Nonaqueous Redox Flow Batteries” J. Am. Chem. Soc. 2016, 137, 15378.
Sevov, C. S.; Brooner, R. E. M.; Chénard, E.; Assary, R. S.; Moore, J. S.; Rodríguez-Lopez, J.; Sanford, M. S., “Evolutionary Design of Low Molecular Weight Organic Anolyte Materials for Applications in Non-Aqueous Redox Flow Batteries” in J. Am. Chem. Soc. 2015, 137, 14465.
Sevov, C. S.; Hartwig, J. F., “Iridium-Catalyzed Oxidative Olefination of Furans with Unactivated Alkenes” in J. Am. Chem. Soc. 2014, 136, 10625-16031.
Sevov, C. S.; Zhou, J.; Hartwig, J. F., “Iridium-Catalyzed, Intermolecular Hydroamination of Unactivated Aliphatic Alkenes with Indoles” in J. Am. Chem. Soc. 2014, 136, 3200-3207.
Sevov, C. S.; Hartwig, J. F., “Iridium-Catalyzed, Intermolecular Hydroetherification of Unactivated Aliphatic Alkenes with Phenols” in J. Am. Chem. Soc. 2013, 135, 9303-9306.
Sevov, C. S.; Hartwig, J. F., “Iridium-Catalyzed Intermolecular Asymmetric Hydroheteroarylation of Bicycloalkenes” in J. Am. Chem. Soc. 2013, 135, 2116-2119.
Sevov, C. S.; Zhou, J.; Hartwig, J. F., “Iridium-Catalyzed Intermolecular Hydroamination of Unactivated Aliphatic Alkenes with Amides and Sulfonamides” in J. Am. Chem. Soc. 2012, 134, 11960-11963.
