Sherwin Singer
Contact Information
Professor
Areas of Expertise
- Physical
Bio
Sherwin Singer received his A.B. 1976 from University of Chicago, and his Ph.D. in 1984, also from the University of Chicago, where he was a National Science Foundation predoctoral fellow working with Karl Freed. He subsequently did postdoctoral studies with David Chandler at University of Pennsylvania, and John Weeks at AT&T Bell Labs. Sherwin Singer has been a faculty member at The Ohio State University since 1987, being promoted to Associate Professor in 1992 and Full Professor in 2003. He was awarded the Camille and Henry Dreyfus Distinguished New Faculty Award in 1986, and the Dozor Fellowship from Ben-Gurion University of the Negev in 1998.
Research Overview
Theoretical Chemistry of Clusters, Films, and Condensed Phases
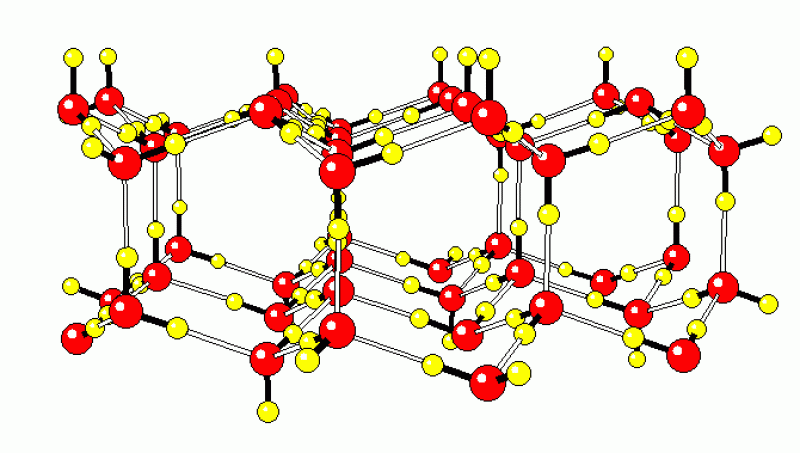
Hydrogen bond order-disorder in ice
Everyone thinks of ordinary ice (ice Ih) as one of the most familiar examples of a crystal. However, the ice cubes in your freezer are not fully ordered crystals. If you took molecular models and built ice crystals like the ones shown below, you would see that there are many Ice Ih different ways to link the water molecules together so that the oxygen atoms form the well-known hexagonal ice Ih lattice. Some of the possible arrangements of H-bonds are shown in the animation. Because there are many different ways to link the molecules in ice with hydrogen bonds, ordinary ice is a disordered material. Other ice phases are also disordered, ice III, V, and VII to name a few. We now believe that if you cool each of the disordered ice phases to a low enough temperature, they will undergo a phase transition to a structure in which a single hydrogen bond pattern is selected. Ice Ih "freezes" into ice XI, ice III into ice IX, ice V into ice XIII, ice VII into ice VIII, and so on. Our work provided the first theoretical calculations that correctly predicted the properties of these phase transitions. This work has been supported by the National Science Foundation.
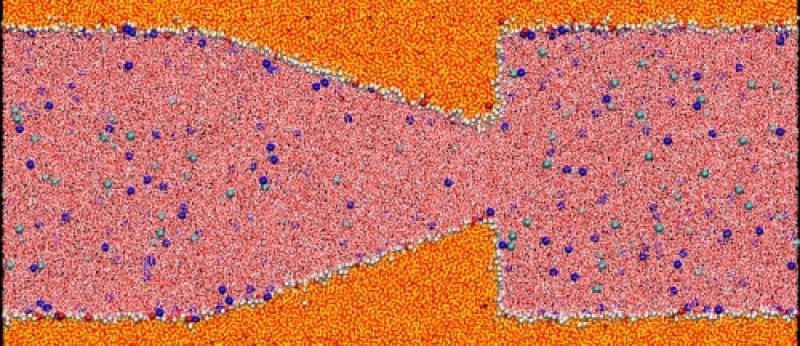
Electroosmotic flow
Working in collaboration with biomedical engineers who design nanoscale devices for drug delivery, we have study flow in a very small channel -- "nanofluidics". We are particularly interested in flow induced by applying an electric field to an electrolyte within nano-scale devices. This is an efficient way to transport fluids within tiny spaces with a high degree of control. In the flow simulation shown on the right, a sodium chloride solution is driven through a "nano-nozzle". The nozzle is made of amorphous silica dioxide (better known as glass). This work has been supported by the National Science Foundation through our participation in the NSEC (Nanoscale Science and Engineering Center) at Ohio State.
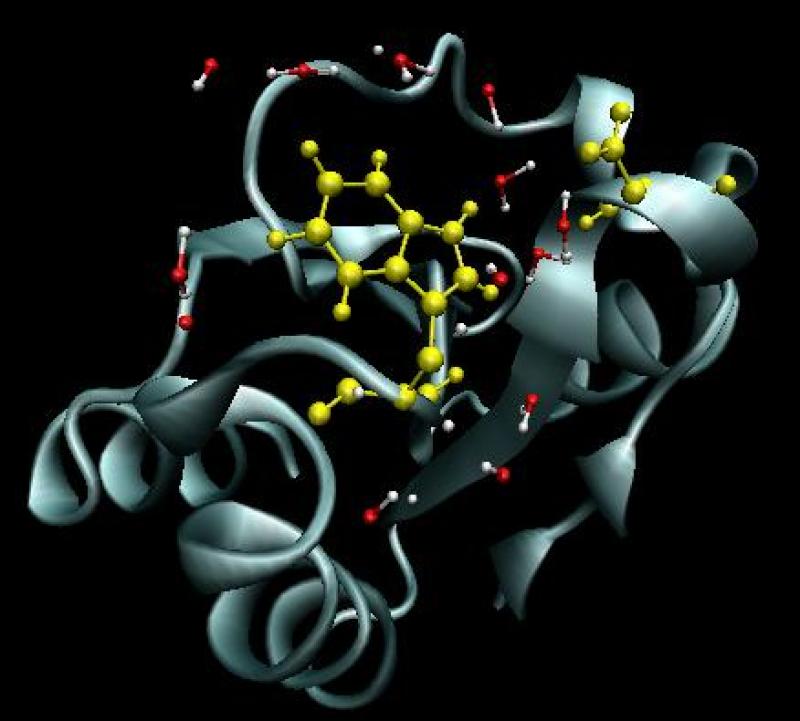
Biomolecular dynamics
Enzyme function requires movement of protein, substrate and water. Without water motion, most proteins lose biological activity. We have analyzed experiments in which a part of the protein absorbs a photon. For example, the indole portion of the tryptophan residue of the enzyme thioredoxin, pictured on the right, can absorb, and later emit, a photon. This alters the charge distribution near the chromophore, and causes both the protein and water to adjust to the new charge distribution. These relaxation processes are reflected in the change in emission wavelength with time (fluorescence Stokes shift). Our calculations provide insight into the contribution of factors like protein flexibility and solvent motion to the observed Stokes shift. Another area of interest is the repair of cyclobutane pyrimidine dimers, the most prevalent form of light-induced damage of genetic material. This work has been funded by the National Institutes of Health.
Materials that spontaneously form patterns
We have developed theories for complex 2-dimensional fluids that spontaneously generate patterns. Our work, like the computer simulations shown on the left, explains the appearance of stripes, bubbles, or intertwined snake-like structures which are experimentally observed in a surprisingly wide variety of physical systems: thin magnetic films, insoluble biomolecules at an air-water interface (Langmuir monolayers, like the layer of lung surfactant that line the surface of alveoli), and adsorbed molecular monolayers on a crystal surface.

Recent Publications
- Antitsa D. Stoycheva and Sherwin J. Singer, ``Stripe melting in a two-dimensional system with competing interactions'', Phys. Rev. Lett. 84(20):4657 (2000).
- Sherwin J. Singer, ``Buckling induced by dilative strain in two- and three-dimensional layered materials'', Phys. Rev. E62(3):3736 (2000).
- Cristian V. Ciobanu, Lars Ojamäe, Isaiah Shavitt and Sherwin J. Singer, ``Structure and vibrational spectra of H+(H2O)8: Is the excess proton in a symmetrical hydrogen bond?'', J. Chem. Phys. 113(13): 5321 (2000).
- Jer-Lai Kuo, James V. Coe, Sherwin J. Singer, Yehuda B. Band, and Lars Ojamäe, ``On the use of graph invariants for efficiently generating hydrogen bond topologies and predicting physical properties of water clusters and ice'', J. Chem. Phys. 114(6):2527 (2001).
- Antitsa D. Stoycheva and Sherwin J. Singer, ``Scaling theory for two-dimensional systems with competing interactions'', Phys. Rev. E64(1):016118 (2001).
- Christopher Callam, Sherwin J. Singer, Todd L. Lowary, and Christopher M. Hadad, ``Computational analysis of the potential energy surfaces of glycerol in the gas and aqueous phases. Effects of level of theory, basis set, and solvation on strongly intramolecularly hydrogen-bonded systems'', J. Amer. Chem. Soc. 123(47):11743 (2001).
- Antitsa D. Stoycheva and Sherwin J. Singer, ``Computer simulations of a two-dimensional system with competing interactions'', Phys. Rev. E65(3):036706 (2002).
- Jer-Lai Kuo and Sherwin J. Singer, ``Graph invariants for periodic systems: towards predicting physical properties from the hydrogen bond topology of ice'', Phys. Rev.E67(1):016114 (2003).
- Jer-Lai Kuo, Cristian V. Ciobanu, Lars Ojamäe, Isaiah Shavitt and Sherwin J. Singer, ``Short H-Bonds and Spontaneous Self-Dissociation in (H2O)20: Effects of H-Bond Topology'', J. Chem. Phys. 118(8):3583 (2003).
- Xinchuan Huang, Hyung Min Cho, Stuart Carter, Lars Ojamäe, Joel M. Bowman, and Sherwin J. Singer, ``Full Dimensional Quantum Calculations of Vibrational Energies of H5O2+'', J. Phys. Chem. 107(37):7142 (2003).
- Hyung Min Cho and Sherwin J. Singer, ``Correlation function quantum Monte Carlo study of the excited vibrational states of H5O2+'', J. Phys. Chem. A108(41):8691 (2004).
- Stephen D. Belair, Joseph S. Francisco and Sherwin J. Singer ``Hydrogen bonding in cubic (H2O)8 and OH·(H2O)7 clusters'', Phys. Rev. A71 (1): 013204 (2005).
- Wei Zhu, Sherwin J. Singer, Zhi Zheng and A. T. Conlisk, ``Electroosmotic flow of a model electrolyte'', Phys. Rev. E71(4): 041501 (2005).
- Wei Zhu and Sherwin J. Singer, ``Monte Carlo simulation of methyl chloride monolayer on the surface of graphite'', Surf. Sci. 579(2-3):141 (2005).
- Sherwin J. Singer, Jer-Lai Kuo, Tomas K. Hirsch, Chris Knight, Lars Ojamäe and Michael L. Klein, ``Hydrogen bond topology and the ice VII/VIII and Ih/XI proton ordering phase transitions'', Phys.Rev.Lett. 94(13):135701 (2005).
- Chris Knight and Sherwin J. Singer, ``Prediction of a phase transition to a hydrogen bond ordered form of ice VI'', J.Phys.Chem. B109(44):21040 (2005).
- Chris Knight, Sherwin J. Singer, Jer-Lai Kuo, Tomas K. Hirsch, Lars Ojamäe and Michael L. Klein, ``Hydrogen bond topology and the ice VII/VIII and Ih/XI proton ordering phase transitions'', Phys.Rev. E73(5):056113 (2006).
- Ali Hassanali, Tanping Li, Dongping Zhong and Sherwin J. Singer,``Lys-trp-lys: Structure and dynamics in solution following photoexcitation'', J. Phys. Chem. B110(21):10497 (2006).
- Chris Knight and Sherwin J. Singer, ``A re-examination of the ice III/IX hydrogen bond ordering phase transition '', J.Chem.Phys. 125(6):064506 (2006).
- A. T. Conlisk and Sherwin Singer, ``Modeling electroosmotic flow in nanochannels.'' BioMEMS and Biomedical Nanotechnology 4:301-330 (2006).
- Ali A. Hassanali and Sherwin J. Singer, ``Static and dynamic properties of the water/amorphous silica interface: a model for the undissociated surface'', J.Computer-Aided Materials Design, 14(1):53 (2007).
- Tanping Li, Ali A. Hassanali, Ya-Ting Kao, Dongping Zhong and Sherwin J. Singer, ``Hydration dynamics and time scales of coupled water-protein fluctuations'', J. Amer. Chem. Soc. 129(11):3376 (2007).
- Chris Knight and Sherwin J. Singer, ``Tackling the problem of hydrogen bond order and disorder'', Physics and Chemistry of Ice (Proceedings of the 11th International Conference on the Physics and Chemistry of Ice), ed., Werner F. Kuhs (Royal Soc. of Chemistry, 2007), p.329.
- Chris Knight and Sherwin J. Singer, ``Theoretical study of a hydroxide ion within the ice-Ih lattice'', Physics and Chemistry of Ice (Proceedings of the 11th International Conference on the Physics and Chemistry of Ice), ed., Werner F. Kuhs (Royal Soc. of Chemistry, 2007), p.339.
- Ali Hassanali and Sherwin J. Singer, ``A model for the water/amorphous silica interface: the undissociated surface'', J. Phys. Chem. B111(38):11181 (2007).
- Tanping Li, Ali A. Hassanali, Sherwin J. Singer, ``The origin of slow relaxation following photoexcitation of W7 in myoglobin and the dynamics of its hydration layer'', J. Phys. Chem. 112(50):16121 (2008).
- Chris Knight and Sherwin J. Singer, ```Hydrogen bond ordering in ice V and the transition to ice XIII'', J. Chem. Phys. 129(16):164513 (2008).
- Chris Knight and Sherwin J. Singer, ``Site disorder in ice VII arising from hydrogen bond fluctuations'', J. Phys. Chem. A113(45):12433 (2009).
- Ali A.Hassanali, Hui Zhang, Chris Knight, Yun Kyung Shin and Sherwin J. Singer, ``The dissociated amorphous silica surface: Model development and evaluation'', J. Comput. Theoretical. Chem. 6 (11):3456 (2010).
- Hui Zhang, Ali A.Hassanali, Yun Kyung Shin, Chris Knight and Sherwin J. Singer, ``The water-amorphous silica interface: Analysis of the Stern layer and surface conduction'', J. Chem. Phys. 134(2):024705 (2011).
